|
Numerous new improvements in the field of cardiopulmonary revival (CPR) have prompted the advancement of many progressed clinical revival advances. Cardiopulmonary revival (CPR) is a lifesaving strategy utilized in various crises including respiratory failure, coronary course sickness (CAD), and heart valve infection. As indicated by the Centers for Disease Control and Prevention (CDC), in 2017, around 365,914 individuals pass on because of CAD with around 18.2 million grown-ups in the U.S. old enough 20 years or more have CAD. Such monstrous rate of heart illnesses has expanded the interest for proper clinical devices. Subsequently, these components can animate development of the automated CPR devices market.
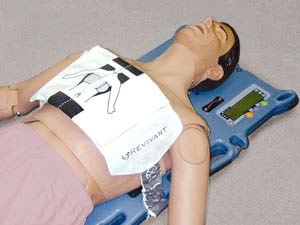
Automated CPR devices help to convey high recurrence and reliable compressions during the cardiopulmonary revival stage. This is basic to accomplishing most extreme results and diminishing the odds of the casualty passing out because of ventricular tachycardia, ventricular fibrillation, or shudder heart musicality. To the extent topographical impact is thought of, North America is by all accounts amassing critical footing in the automated CPR devices market. This development can be ascribed to high geriatric populace and mechanical headways in clinical devices. Despite what is generally expected, Europe is a key area enrolling promising signs because of an ideal administrative structure and high discretionary cashflow. As of late, in June 2020, Royal Philips got the U.S. Food and Drug Administration (FDA) premarket endorsement (PMA) for its HeartStart FR3 and HeartStart FRx automated outside defibrillators (AEDs). Read More : https://bit.ly/3yf3fYB
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2021
Categories |
 RSS Feed
RSS Feed
